RHU OPERANDI
University-Hospital Research project
Optimisation and imProved Efficacy of targeted RAdioNuclide therapy in Digestive cancers by Imagomics
About
The University-Hospital Research projects (RHU) are excellence programmes supported by the future investment plan and selected
by the The French National Research Agency (ANR).
They aim to support innovative and large-scale research projects in the field of health.
Scientific coordinator
Professor Valérie Vilgrain
Department of Radiology
Beaujon Hospital
AP-HP. Nord -
Université Paris Cité
Targeted diseases
The Hepatocellular Carcinoma (HCC)
and the Gastro-Entero-Pancreatic
Neuro-Endocrine Tumors
(GEP-NETs)
Total project duration
5 years
2022 > 2027
- Digestive Oncology.
- Hepatocellular Carcinoma.
- Neuroendocrine Tumour.
- Targeted Radionuclide Therapy.
- Radiopharmaceuticals.
- Biomarker.
- Medical Imaging.
- Imagomics.
- PET-MRI.
- Artificial Intelligence.
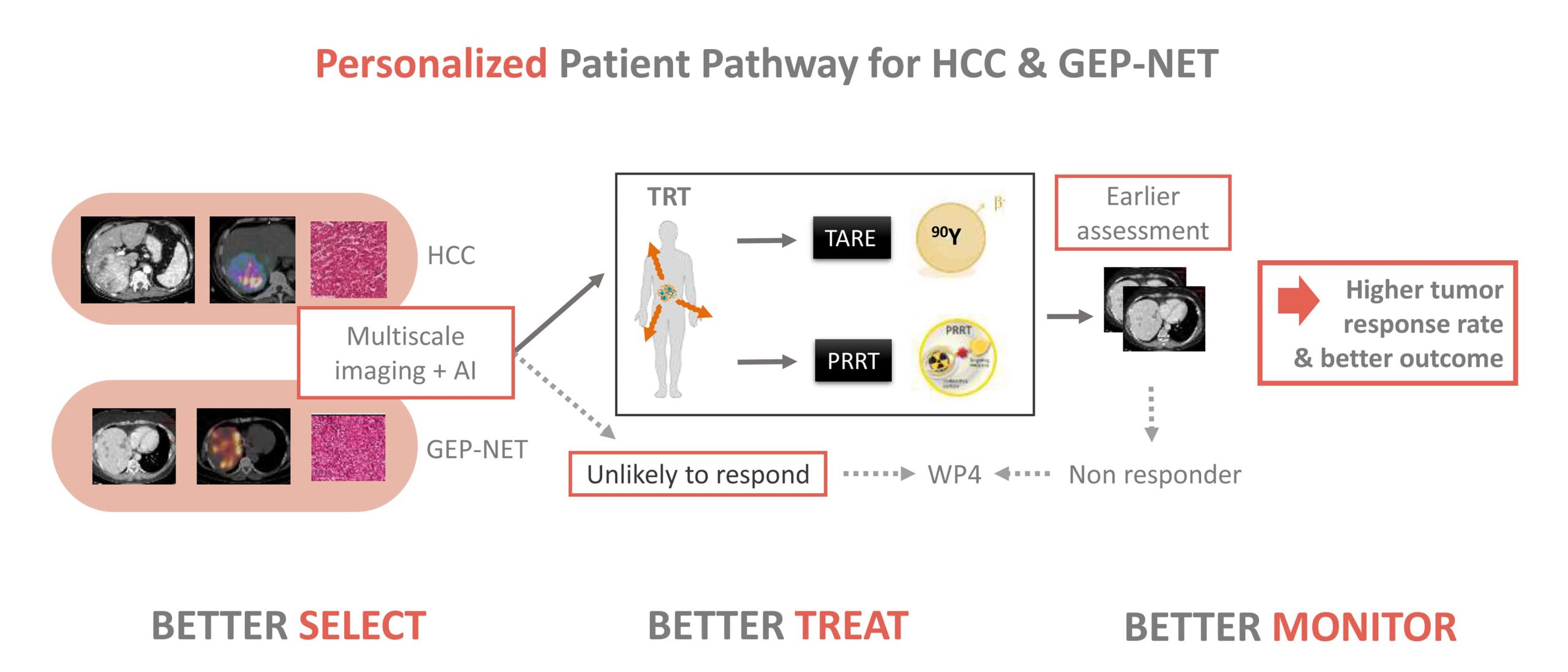
The OPERANDI project (Optimisation and imProved Efficacy of targeted RAdioNuclide therapy in Digestive cancers by Imagomics) aims to address unmet clinical needs in the current management of advanced stage digestive tumours treated with targeted radionuclide therapy (TRT) by exploring new opportunities provided by imaging-based artificial intelligence (AI) and data augmentation, simultaneous PET-MRI imaging, and novel approaches to increase TRT efficacy (genomic profiling, radiopotentiators, and new radionuclides).
TRT is a novel and most promising therapeutic approach in hepatocellular carcinoma (HCC) and gastroenteropancreatic neuroendocrine tumours (GEP-NET) and is the most suitable therapeutic option in intermediate/advanced HCCs and inoperable metastatic GEP-NETs. However, currently there is a “one-fits-all” approach rendering patient stratification and monitoring of therapy response inefficient. This leads to loss of precious time for non-responders which could benefit otherwise much earlier from treatment changes whereby impacting outcome.
HCCs and GEP-NETs share striking common features making their distinction even for pathologists sometimes very difficult. Consequently, they are both considered within the OPERANDI project because any exclusion of any one of them would contradict clinical reality.
HCC is one of the most common malignant tumours, considered the sixth most common cancer and the third major cause of cancer-related death. Its worldwide incidence is expected to increase in the future [1]. While GEP-NETs were considered rare tumours, their incidence has been increasing over the last few decades, and the prevalence of GEP-NETs is higher than the combined prevalence of other more common gastrointestinal cancers, including esophageal cancer, gastric adenocarcinoma, and pancreatic adenocarcinoma [2]. Additionally, prognosis is related to liver involvement for both tumours types, they show marked intertumour and intratumour heterogeneity at the microscopic and molecular level, and presently there are no reliable imaging markers gauging response to therapy.
The OPERANDI project has three main objectives to globally address the challenges of TRT in these tumours:
- To identify predictive and early markers indicative of TRT effectiveness: two large retrospective cohorts of HCC and GEP-NET patients will be utilized to uncover relevant predictive signatures within the morphological, functional, and molecular imaging data using novel imaging-based AI approaches. Particularly, we will benefit from the concept of data augmentation to render the proposed algorithms statistically significant. In a prospective study motion compensated simultaneous PET-MRI will provide unprecedented image quality for extended lesion characterization and imaging biomarker quantification. This imaging-based “omics” approach (imagomics) will firstly allow to select patients that are most likely to respond to TRT, and secondly enable to gauge response to therapy at sooner timepoints. Overall, this will improve the patient pathway and allow a swift transfer to industry, a key aspect when TRT will be deployed to other cancer types.
- To increase TRT efficacy: radiotherapy induces DNA damage and DNA repair capability of tumour cells whereby probably conditioning their resistance. We will develop predictive genomic signatures of TRT sensitivity in human cohorts and explore the mechanisms of resistance in vitro to pinpoint the key pathways which in return could be targeted in a second step. TRT radiopotentiators targeting these pathways will be evaluated in vitro on HCC and GEP-NET cell lines and organoids, and their synergic action will be investigated using unique DNA repair functional assays. The best candidates will be tested in orthotopic PDX models. This will help to select from various molecules the most promising ones and shed light on the key DNA repair systems involved in TRT sensitivity/resistance to further guide the development of new companion molecules.
- To develop new TRT approaches: new target proteins such as PSMA and CAIX will be assessed in HCC and/or GEP-NET highlighting the translation between the two tumours. We expect that those novel concepts allow us to target aggressive and refractory tumours currently not responding to TRT. New theranostic approaches will be evaluated in clinical trials. Radiolabeled agents will be evaluated with α-emitters in GEP-NET enabling higher cytotoxicity and spatial localization in refractory/relapsing tumours. Toxicity and efficacy of α-TRT will be assessed in animal models.
Collectively, OPERANDI will impact: i) advanced stage cancer patients who require care at the highest medical level using novel signatures and imaging biomarkers; ii) patient management with a holistic approach via PET-MRI guided therapy and imaging-based AI algorithms for prediction; iii) novel pathophysiologic therapy pathways and identification of new targets for existing radionuclides; and finally, pharmacologic research that facilitates rapid transfer.
The RHU OPERANDI is structured into eight complementary workpackages (WP), aiming to implement:
- Clinical cohorts (WP0). The leader of this WP0 is Dr. Axelle Dupont, Unité de recherche clinique Paris Nord Val-de-Seine (URC PNVS), Direction de la Recherche Clinique et de l’Innovation (DRCI), Bichat-Claude Bernard Hospital (AP-HP).
- To integrate data to provide artificial intelligence algorithms based on predictive and prognostic imaging (WP1). The leader of this WP1 is Pr. Stéphanie Allassonière, Université Paris Cité.
- To propose a totally innovative and holistic approach via guided therapy simultaneously by PET-MRI (WP2). The leader of this WP2 is Pr. Ralph Sinkus, Inserm U1148 LVTS (Laboratory for Vascular Translational Science).
- To decode the molecular events associated with tumor resistance/response to treatments (WP3). The leader of this WP3 is Pr. Jérôme Cros, Inserm U1149 CRI (The Inflammation Research Center).
- To evaluate new strategies for internal radiation therapy (WP4). The leader of this WP4 is Pr. Françoise Kraeber-Bodéré, Department of Nuclear Medicine (University Hospital of Nantes).
- To validate prospectively these new strategies of internal radiation therapy (WP5). The leader of this WP5 is Dr. Louis De Mestier, Department of Gastroenterology and Pancreatology, Beaujon Hospital (AP-HP).
Two workpackages are also dedicated to project management (WP6) and to disseminating knowledge and project results (WP7). The leader of the WP7 is Pr. Maxime Ronot, Department of Radiology, Beaujon Hospital (AP-HP).








